No products in the cart.
Medical Devices
Showing 1–30 of 106 resultsSorted by popularity
- Medical Suction Accessories
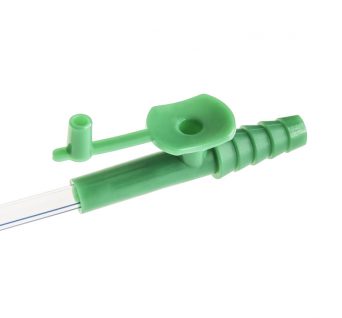
Adult Yankauer Suction Handle
For use with medical suction units
The Adult Yankauer suction tube is an indispensable part of a first responder’s tool-kit and a staple among hospital units.
It is used as an oral suctioning tool in medical procedures. It attaches directly to a pump and is ideal for first responders, first-aid environments and minor surgeries.
The firm plastic suction tip is disposable and features a large opening surrounded by a bulbous head.
SKU: AN170002 - Oxygen Therapy Consumables & Accessories
Oxygen Nasal Cannula, Adult Size
Super soft, flexible nasal prong inserts for added patient comfort. Kink resistant star lumen tubing
Nasal Oxygen Cannulas are made from non-toxic medical grade PVC and are designed to simplify oxygen administration and
are super soft and flexible to maximise patient comfort.For superior performance and patient satisfaction, choose our Nasal Oxygen Cannula with 2.1m clear tubing. It combines comfort, durability, and functionality to support effective oxygen therapy.
SKU: AN070002NS - Nebuliser Accessories
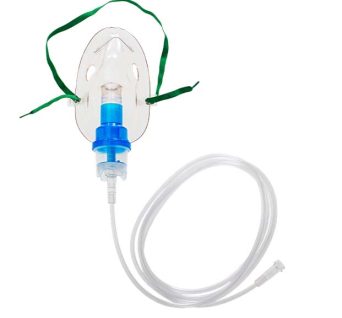
Nebuliser Mask, Adult
Designed to deliver medication in the form of a fine mist, or aerosol, directly to the lungs connected to compressor nebuliser.
The mask is connected to the air compressor, via the nebulizer bowl, by 2 meters of clear latex-free tubing attached with a swivel connection which permits some patient movement. This is typically used for patients with respiratory conditions such as asthma, COPD, bronchitis, or other respiratory infections that require direct lung treatment.
SKU: AN081001NS - Oxygen Therapy Consumables & Accessories
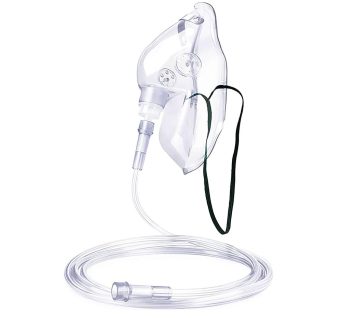
Oxygen Mask With Tubing
With elastic straps, adjustable aluminum nose clip to ensure secure and comfortable fit. Made latex free from medical grade PVC.
For delivering effective aerosol and oxygen therapy, the Adult-sized oxygen mask with Tubing allows for needed medications, oxygen, or even humidity to be placed in a patient’s respiratory tract and lungs.
Perfect for hospitals, clinics, and home care. Ensure optimal oxygen delivery with a mask that prioritizes patient comfort and clinical effectiveness. Order now to stock up on this essential medical supply!
SKU: AN061003NS - Sphygmomanometers
Aneroid Sphygmomanometer Two Handed
Aneroid sphygmomanometer consists of an inflatable cuff, a measuring unit and bulb and valve
Used to measure blood pressure, composed of an inflatable cuff to collapse and then release the artery under the cuff in a controlled manner and an aneroid manometer to measure the pressure
The simple, classic design is easy to use and has been relied upon by healthcare professionals for many years.
SKU: 5189 - CPAP Masks
DJMed Nasal Pillow CPAP Mask
Type 1 nasal pillows mask comes complete with a frame, a selection of nasal pillows, and adjustable elastic headgear
Pillow masks provide a seal around each nostril with minimum contact to the face.
Nasal pillows CPAP masks are small, compact and lightweight. They’re good for people who toss and turn in their sleep or who feel claustrophobic with a larger sleep apnea mask. Also, if you have a beard or moustache, this could be a good choice. You can easily read or watch TV while wearing a nasal pillows mask, even with glasses on.
Nasal pillow CPAP mask can be used with any brand of CPAP (APAP, Fixed Pressure CPAP or BiPAP) machine except for the certain travel CPAP machines. Compatible mask for all CPAP machines that attaches to the machine via 22mm hose connector.
SKU: 211217 - Stethoscopes
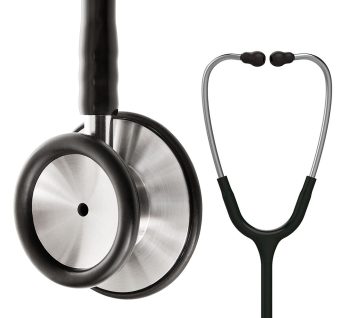
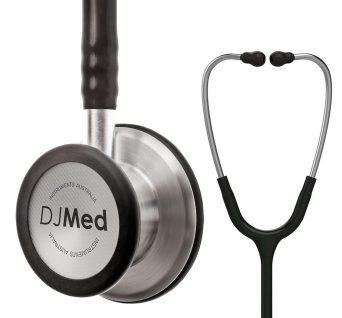
Diagnostic Stethoscope, Dual Head
Classic dual head, a bell and floating, tunable diaphragm design, made with precision stainless steel
Dual head stethoscope is a quality diagnostic instrument used for the auscultation of heart, lung and bowel sounds.
Hard-working clinical tool with the acoustic sensitivity and versatility you can rely on.
SKU: 505150 - CPAP Masks
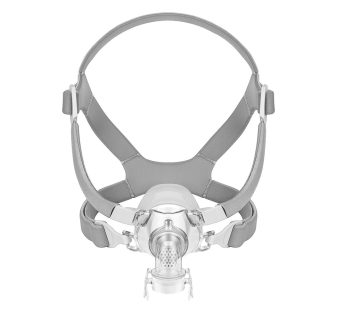
DJMed Full Face CPAP Mask
DJMed Type 2, full face CPAP mask let you breathe with your mouth open while you sleep
Full face CPAP masks cover your mouth and nose. They’re often recommended for people who need a higher pressure as well as those who prefer to breathe through their mouth and/or have trouble breathing through their nose. If you like to sleep on your back, this could be the sleep apnea mask for you.
DJMed CPAP Masks can be used with any brand of CPAP (APAP, Fixed Pressure CPAP or BiPAP) machine except for the certain travel CPAP machines. Compatible mask for all CPAP machines that attaches to the machine via 22mm hose connector.
SKU: n/a - Pulse Oximeters
Pulse Oximeter, Blood Oxygen Monitor
Meet One of the World’s Most Advanced Pulse Oximeters
Pulse Oximeter measures your Oxygen Saturation (Sp02) & Pulse Rate.
The oximeter is portable and ideal for monitoring respiratory conditions; pre/post-exercise and pre/post-operative conditions.
SKU: 200201 - Nebuliser Accessories
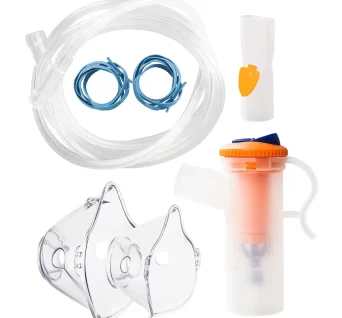
Nebuliser Bowl Kit
Nebuliser kit with masks, nebuliser bottle and tubbing.
An accessory set, which includes masks, bottles and tubings for those patients undergoing nebuliser therapy at home. It comes in with two sizes for the mask, one for adult use and another for pediatric use.
Masks are adjustable to provide for a perfect fit for the user.
SKU: 911021 - Oxygen Therapy Consumables & Accessories
Nasal Oxygen Cannula Ear Cushions
Tube cushions are soft, closed-cell foam cushions that slide over the head or face tube of an oxygen cannula
This will provide a cushion to reduce chafing or pressure on the ear area. It is Latex-free. Sold as a pair (2pcs)
SKU: 1016-0 - Nebulisers
Homecare Compressor Nebuliser
Stylish compressor nebuliser perfect for facility and homecare use
DJMed homecare nebuliser from DJMed is built to treat a range of respiratory conditions: COPD, asthma, and other lung diseases.
This device produces a fine mist of droplets that contain active medication delivered into the lungs to help your condition.
The nebuliser is small, strong, and simple to use. With proper use and care, this device will provide you with many years of treatment.
Order now for a FREE delivery!
SKU: 9100023 - Nebulisers
Clinical Compressor Nebuliser
Clinical compressor nebuliser with accessory storage compartment
For patients with a respiratory illness, such as asthma, nebulisers offer a quick and effective way to find relief from their symptoms.
With the use of a nebulizer, patients can inhale their prescribed medication directly into the lungs, giving them fast relief from inflammation — and allowing them to breathe easier.
Order now for a FREE delivery!
SKU: 910024 - TENS & EMS Accessories
TENS Electrode Pads, Square, 50x50mm, White
Self-adhesive, re-usable, long Life, 50×50 mm, 2mm plug, 10pcs pads included
Reusable electrodes with white super soft cotton (allow attachment to skin smoothly) backing and premium adhesive gel.
Popular size, bio-compatible, long life dry out Resisting, durable, comfortable pads that gently adhere to your skin. Perfect for TENS and EMS.
SKU: 300501 - TENS Machines
Digital Therapy, Rechargeable TENS Machine
Dual channel, digital electrotherapy pain relief EMS & TENS unit with USB rechargeable lithium battery.
Digital TENS Machine is an easy to use machine that offers non-invasive, natural pain relief without the use of any drugs or chemicals.
SKU: 300051 - Stethoscopes
Classic Stethoscope
Hard-working, dependable and sensitive Classic stethoscope is perfect for assessing children or adults
Tunable, adjustable frequency, floating diaphragm allows tuning to enhance high- and low-frequency response in both the adult and pediatric diaphragms.
Flexible PVC tubing provides excellent sound conduction, and the sleek design of this product offers professional styling along with superior clinical performance.
SKU: n/a - CPAP Masks
DJMed Nasal CPAP Mask
DJMed Type 3, nasal CPAP mask is ideal if you breathe through your nose while you sleep
Nasal CPAP masks cover your nose from the bridge to the area at the top of your lip. They deliver an indirect airflow and are a good choice if you need higher pressure. If you’re a restless sleeper you may prefer this type of sleep apnea mask.
DJMed CPAP Masks can be used with any brand of CPAP (APAP, Fixed Pressure CPAP or BiPAP) machine except for the certain travel CPAP machines. Compatible mask for all CPAP machines that attaches to the machine via 22mm hose connector.
SKU: n/a - TENS & EMS Accessories
TENS Electrode Pads, Square, 50x50mm, Black
Self-adhesive, re-usable, long Life, 50×50 mm, 2mm plug, 10pcs pads included
Reusable electrodes with black super soft cotton (allow attachment to skin smoothly) backing and premium adhesive gel.
Popular size, bio-compatible, long life dry out Resisting, durable, comfortable pads that gently adhere to your skin. Perfect for TENS and EMS.
SKU: 300502 - Stethoscopes
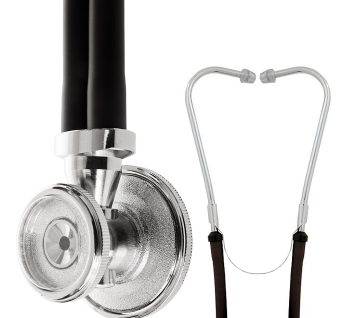
Sprague Rappaport Stethoscope
Affordable, multi-purpose stethoscope with excellent acoustics designed for clinical use.
The legendary design features of the Sprague have made it one of the most popular stethoscopes in history, traditionally trusted to detect faint heart sounds and murmurs.
Unique five-in-one stethoscope that eliminates the need for different stethoscopes for infants, children, and adults.
SKU: 3523 - Nursing Alarms & Pagers
Two Call Buttons and Pager Kit for the Elderly
2 x Call Buttons – Mobile Pager – Easy One Button Use – 50m Call Distance
Caregiver Wireless Pager, a Nurse Call System
A Caregiver wireless pager comes with two buttons, one could be placed in the bathroom and one to hang around your neck, upstairs and one down stairs or wherever one may wish.
Wireless nurse call system comes with a mobile pager for the carer to carry in their pocket or to clip onto their belt. With one simple press of a button the resident is able to call the carer for assistance.
The wireless pager has a range is significant at 50m allowing the caregiver freedom of movement and the resident a feeling of safety.
SKU: 2014 - TENS Machines
Pro Therapy TENS Machine
Dual channel, professional high frequency, wide pulse width max. intensity TENS unit with a safe-storage case.
TENS machine offers a proven and effective pain relief together with a portable travel size. TENS machine offers program modes that are preset for relaxing variable massage and various electronic pulse modes to help relieve acute and chronic pain.
TENS machine is easy to use with pre-set automatic programs for suit parts of your body such as back, shoulders and more. With an easy visual body icon on the LCD display that displays massage style, intensity and time remaining.
SKU: 300050 - CPAP Supplies
Classic CPAP Hose Tubing
Standard 19mm tubing with 22mm connection
Flexible CPAP tubing has the industry standard 22mm connection and a standard length of 1.8cm (6′), making it compatible with almost all equipment. The flexible and light tube reduces the pull on your mask so you can sleep comfortably.
SKU: 211310
Medical Devices
In Australia, a medical device is defined and regulated by the Therapeutic Goods Administration (TGA), which is the regulatory authority responsible for ensuring the safety and efficacy of therapeutic goods, including medical devices. The TGA defines a medical device as follows:
"A medical device is any instrument, apparatus, appliance, software, material, or other article intended to be used for a medical purpose and does not achieve its primary intended action by pharmacological, immunological, or metabolic means in or on the human body, but which may be assisted in its intended function by such means."
This definition encompasses a wide range of products used in healthcare, including but not limited to:
Diagnostic Equipment: Such as blood glucose monitors, X-ray machines, and MRI scanners. Surgical Instruments: Such as scalpels, forceps, and surgical implants like joint replacements or pacemakers. Medical Implants: Devices that are surgically placed inside the body, such as artificial joints, heart valves, or stents. Diagnostic Software: Software used for medical diagnosis, monitoring, or treatment planning. Personal Protective Equipment (PPE): Certain medical devices used for infection control and safety, including surgical masks, gloves, and face shields. In vitro Diagnostic (IVD) Devices: Devices used for testing samples outside the body, such as pregnancy tests, blood glucose test strips, and laboratory equipment. Assistive Devices: Products designed to assist individuals with disabilities or medical conditions, such as hearing aids, wheelchairs, and walking aids. Dental Equipment: Dental instruments, X-ray machines, and dental implants are also considered medical devices. Wound Care Products: Dressings, bandages, and wound management devices.
Medical devices in Australia are classified into various risk classes (Class I, Class IIa, Class IIb, Class III, and Active Implantable Medical Devices), and the level of regulatory scrutiny and requirements vary depending on the classification. The TGA assesses and regulates medical devices to ensure they meet safety, quality, and performance standards before they can be legally marketed and used in Australia.
Manufacturers of medical devices are required to comply with regulatory requirements, including registering their products with the TGA and maintaining appropriate records. Healthcare professionals and consumers can also access information about registered medical devices on the TGA's public database.
It's important to note that medical devices play a critical role in healthcare, and their regulation is essential to ensure patient safety and the effectiveness of these products. If you have questions about a specific medical device or its regulatory status in Australia, it is advisable to consult with healthcare professionals or refer to the TGA's official resources.
What is an example of a medical device?
Certainly, here are some examples of medical devices:
Blood Pressure Monitor: This device is used to measure a patient's blood pressure and is often used at home or in clinical settings. Thermometer: A common medical device used to measure body temperature, which is essential for diagnosing fever and monitoring a patient's health. Insulin Pump: Used by people with diabetes, an insulin pump delivers a controlled dose of insulin to regulate blood sugar levels. Pacemaker: An implantable medical device used to regulate the heart's rhythm by sending electrical impulses to the heart muscles. Ventilator: A machine that assists patients with breathing when they are unable to do so adequately on their own, often used in intensive care units. Infusion Pump: Used to deliver fluids, medications, or nutrients directly into a patient's bloodstream in a controlled manner. X-ray Machine: A medical imaging device that uses X-rays to create images of the body's internal structures, helping diagnose and monitor various conditions. Blood Glucose Monitor: A device used by people with diabetes to measure their blood glucose levels and manage their condition. EKG/ECG Machine: A device that records the electrical activity of the heart, commonly used to diagnose heart conditions. Ultrasound Machine: Medical imaging equipment that uses sound waves to create real-time images of internal organs and tissues.
These examples illustrate the diversity of medical devices and their crucial roles in healthcare, from diagnostic tools to therapeutic and life-supporting devices. The classification and regulation of medical devices vary by country and region to ensure their safety, effectiveness, and quality of use in healthcare settings.